A Decade of Hyperbaric Oxygen Therapy (HBOT) for Cancer Patients
In 2015, Nature Works Best Medical Clinic installed its first two hyperbaric (HBOT) chambers for use with cancer patients and others. This was a daring act for a cancer clinic, because the very large and powerful coalition of chemotherapy / pharmaceutical / insurance industries and their affiliated media already frowned on this clinic’s prior decade of using anti-cancer nutrients to eliminate cancer. But then we began adding yet another low cost and harmless treatment of cancer by simply pressurizing room air for oxygen delivery to tissues. Anti-naturopathic media editorials from previous decades gradually gave way to silence, as there were no negative reports that patients reported from naturopathic treatments.
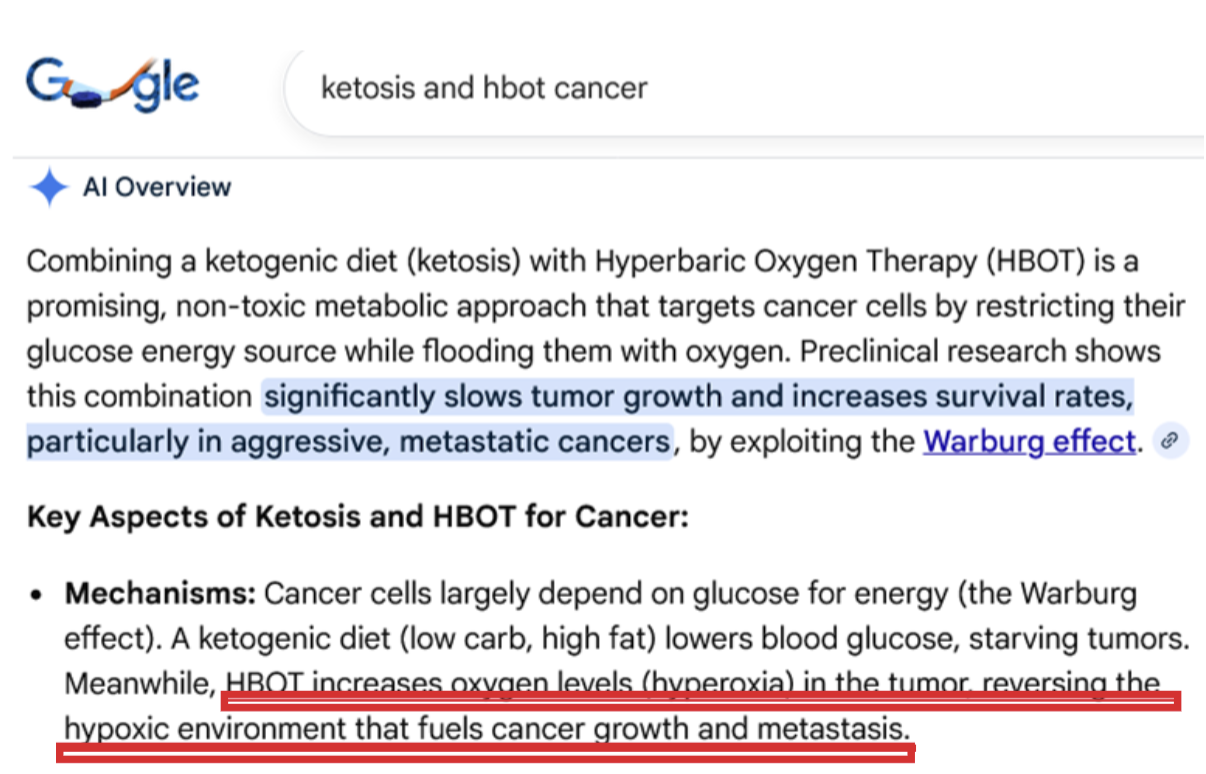
Now, times have changed so much that even Google Gemini AI admits of HBOT effects against cancer.
HBOT versus cancer – main function
Hyperbaric oxygen therapy (HBOT) saturates tumors with oxygen. This is thought to reverse the cancer-promoting effects of tumor hypoxia. This article will discuss that mechanism and related anti-cancer activity at the level of molecules and cells.
But first, the physics of hyperbaric oxygen therapy is needed.
HBOT mechanics
Let’s look at some physics of hyperbaric oxygen therapy (HBOT). The chamber is triple-zipped up tightly, because room air pressure is much lower than in the hyperbaric chamber, so we have to keep those airspaces separate. As the pressure differentiates, at the beginning and at the end of an HBOT session, pressurization and depressurization is done only gradually, never suddenly. This process happens in reverse even when you take a flight, which is opposite in (equivalent) altitude and opposite in pressure.
Here in Tempe, we are at about 1,200 feet above sea level. So in Tempe we are at 97 kilopascal of atmospheric pressure. The HBOT adds 31 kilopascal (kPa) of pressure to that, to total 128 kPa pressure.
Pressure in a hyperbaric chamber is additive: the pressure at sea level (or higher) plus the added pressure in the chamber. Therefore, an inflated hyperbaric chamber delivers 1.3 ATM = 1.0 ATM at sea level plus 0.3 ATM added pressure from the compressor forcing room air into a sealed chamber.
That is, 97 kPa + 31 kPa brings us to 128 kPa inside the chamber, which is about 1.3 atmospheres or 1.3 ATM, sometimes written as 1.3 ATA. We can see this in the table below. 131 kPa = 1.3 ATM.
Biophysics and medicine reveal the advantages of increased pressure
Hyperbaric therapy has been helpful to people in many ways. Let’s start with a brief summary of the physics involved. Increased pressure does the following:
This means that under higher pressure, more of the oxygen that you inhale becomes available to even the farthest reaches of the body. Every cell in your body is no more than a fingernail’s thickness away from the nearest blood vessel, and the increase in dissolved oxygen is just as ubiquitous.
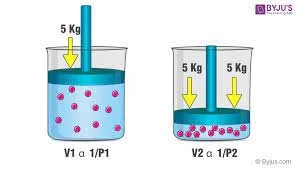
Henry’s Law is described here, from https://www.newleafeugene.com/how-it-works
Here is an explanation of Boyle’s Law from https://www.oxyhealth.com/hyperbaric_therapy.html
The difference between those two laws for our purposes is that Henry’s law explains the arrival and dissolving of oxygen into blood as dependent on pressure, whereas Boyle’s law explains the increased amount of oxygen (more and smaller oxygen molecules) that the blood can absorb as a function of pressure.
Here are all three laws that govern the physics of hyperbaric chambers:
The bottom line of the biophysics of hyperbaric oxygen therapy (HBOT)
The oxygen we breathe in from room air arrives to every part of the body, more efficiently and in greater volume, while under pressure. There is no need to add supplemental oxygen to achieve this effect.
Regarding HBOT therapy, “its primary effect is due to the increased ambient pressure itself.” https://www.ncbi.nlm.nih.gov/books/NBK448104/
That means that no added oxygen is needed, in order to get the benefits of increased oxygen in the tissues of the body. The article at the above link discusses this in detail.
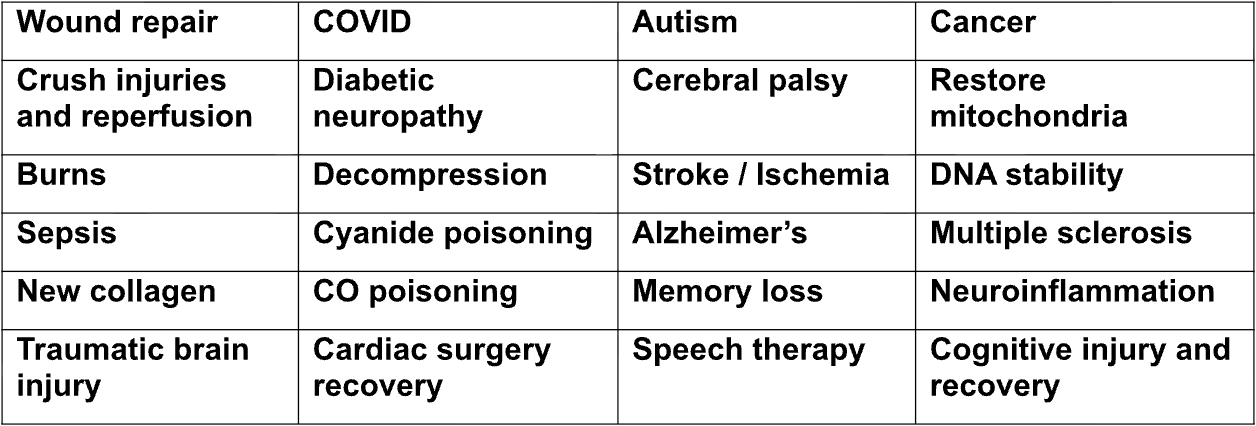
What are the health advantages of ↑pressure→↑oxygen?
Here are some of the many health conditions that hyperbaric therapy has been helpful in treating.
More information on various health conditions treated successfully by HBOT can be found at:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7564723/
Here is how HBOT is thought to work against cancer
from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8465921/
M Ortega, O Fraile-Martinez. A general overview on the hyperbaric oxygen therapy: applications, mechanisms and translational opportunities. Sep 2021. Medicina 57 (9).
HBOT and Cancer
“Cancer is a complex entity which encompasses a broad spectrum of unique pathologies that share the following hallmarks: Immune system evasion, tumor-promoting inflammation, genome instability, enabling replicative immortality, activating invasion and metastasis sustaining proliferative signaling, evading growth suppressors, resisting cell death, inducing angiogenesis, and metabolic reprogramming [156]. Tumor-hypoxia plays a central role in many of these carcinogenic features, promoting an aggressive phenotype besides limit the effectiveness of radiotherapy, chemotherapy, and immunotherapy thereby worsening prognosis in the oncological patients [157]. Thus, targeting tumoral hypoxia and its downstream effectors have been proposed as a potential therapeutical approach in cancer management [158,159,160]. In this line, accumulating evidence supports the role of HBOT in the inhibition of tumor growth and therapy success, by three main mechanisms: (1) By limiting cancer-associated hypoxia, (2) through the generation of ROS and RNS and (3) restoring immune function [161]. Actual investigations show the promising role of HBOT in a wide variety of malignancies, including breast cancer, prostate cancer, head and neck cancer, colorectal cancer, leukemia, brain tumors, cervical cancer and bladder cancer [162]. Main applications derived from HBOT in oncology may be (a) As part of the treatment (b) as a radiotherapy adjuvant and (c) as a chemotherapy adjuvant [163].”
156. Hanahan D., Weinberg R.A. Hallmarks of Cancer: The next Generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [PubMed] [CrossRef] [Google Scholar]
157. Muz B., de la Puente P., Azab F., Azab A.K. The Role of Hypoxia in Cancer Progression, Angiogenesis, Metastasis, and Resistance to Therapy. Hypoxia. 2015;3:83. doi: 10.2147/HP.S93413. [PMC free article] [PubMed] [CrossRef]
158. Ortega M.A., Fraile-Martínez O., Asúnsolo Á., Buján J., García-Honduvilla N., Coca S. Signal Transduction Pathways in Breast Cancer: The Important Role of PI3K/Akt/MTOR. J. Oncol. 2020;2020:9258396. doi: 10.1155/2020/9258396. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
159. Jing X., Yang F., Shao C., Wei K., Xie M., Shen H., Shu Y. Role of Hypoxia in Cancer Therapy by Regulating the Tumor Microenvironment. Mol. Cancer. 2019;18:157. doi: 10.1186/s12943-019-1089-9. [PMC free article] [PubMed] [CrossRef]
160. Dhani N., Fyles A., Hedley D., Milosevic M. The Clinical Significance of Hypoxia in Human Cancers. Semin. Nucl. Med. 2015;45:110–121. doi: 10.1053/j.semnuclmed.2014.11.002. [PubMed] [CrossRef] [Google Scholar]
161. Kim S.W., Kim I.K., Lee S.H. Role of Hyperoxic Treatment in Cancer. Exp. Biol. Med. 2020;245:851–860. doi: 10.1177/1535370220921547. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
162. Moen I., Stuhr L.E.B. Hyperbaric Oxygen Therapy and Cancer—A Review. Target. Oncol. 2012;7:233–242. doi: 10.1007/s11523-012-0233-x. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
163. Stępień K., Ostrowski R.P., Matyja E. Hyperbaric Oxygen as an Adjunctive Therapy in Treatment of Malignancies, Including Brain Tumours. Med. Oncol. 2016;33:101. doi: 10.1007/s12032-016-0814-0. [PMC free article] [PubMed]
The following Google Gemini reply was given to the query “ketosis and hbot cancer”
How Nature Works Best Cancer Clinic uses HBOT against cancer
We have had a longstanding challenge and responsibility at Nature Works Best Clinic. Cancer cells and normal cells are more similar to each other than microbes and normal cells. Therefore, the immune system does not always succeed in targeting and eliminating cancer, and it does a better job against microbes. Therefore, our responsibility is not only to help tag cancer for immune system destruction, but also to find and exploit all of the ways that cancer differs from normal cells, in order to use therapies that nourish normal cells while destroying cancer cells. That is our mission and purpose at the clinic.
One of those cell differences is that normal cells thrive in an oxygen rich milieu (either normoxia or hyperoxia), whereas cancer cells thrive in an oxygen-poor milieu (hypoxia).
Tumors have less than 40% of the oxygen of normal tissues, even more pronounced in smaller tumors and micrometastases.[1] As cancer outgrows its blood supply and therefore oxygen supply, the resulting hypoxia makes tumors more aggressive, in that they tend to become immortal, they tend to build more local blood vessels (angiogenesis), and they are more likely to metastasize. These processes result in worsened patient outcomes.[2][3] The most hypoxic areas of tumors tend to be more resistant to chemotherapy, and tend to harbor cancer stem cells, which have the capacity to re-grow cancer after chemotherapy treatment.[4]
This activity is mediated by a molecule hypoxia-inducible factor 1-alpha. When HIF-1-alpha increases, the above problems increase.[5] Also DNA damage and drug resistance result.[6]
Hypoxia also leads to a low pH environment, also known as acidosis. This process increases the chaos of the genetic alterations. As a result, chemotherapy that was chosen following biopsy, based on genetic patterns observed on histological analysis, are now rendered obsolete for that patient’s tumor burden. The genetics of the tumor have simply changed too fast for chemotherapy regimens to keep up.
Common chemotherapy drugs, cisplatin, carboplatin and doxorubicin are defeated by hypoxia. Here are some of the chemotherapy drugs rendered useless by this hypoxic process in cancer cells.
Table from X Jing, F Yang, et al. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Nvo 2019. Mol Cancer. 18 (157). https://pmc.ncbi.nlm.nih.gov/articles/PMC6844052/#CR16
Therefore, a reversal of hypoxia, by using hyperbaric chambers, should be welcomed by chemotherapy oncologists, as a way to make their drugs’ effects more durable.
Chemotherapy is not advocated or used by our clinic. Rather, our treatments focus on high-dose intravenous nutrients chosen for their anti-cancer effects, and described in our Cancer & Biochemistry videos, https://natureworksbest.com/cancer-biochemistry
Hypoxia in cancer cells indirectly causes reactive oxygen species (ROS) to accumulate in and in the vicinity of cancer cells.[7] This sets up a vulnerability, and then when our nutrient treatments arrive to cancer cells, that IV treatment exploits that vulnerability to kill cancer cells. Here is how that happens: Chen, Espey, et al. showed in 2005 that because normal cells have the enzyme catalase, but cancer cells lack that enzyme, high dose intravenous vitamin C leaves more ROS in the extracelleular fluid surrounding cancer cells, but not normal cells. That is when the already metabolically fragile cancer cells die from the intolerably high ROS in their immediate tumor microenvironment.[8] Yet all this happens without harming normal cells, because normal cells have the catalase enzyme, and are therefore able to break those ROS down to the benign substances oxygen and water.
Another way that HBOT benefits normal cells while attacking cancer cells is this: Angiogenesis, the generation of new local blood vessels, makes cancer more aggressive and more dangerous. So we want to stop that process in cancer cells. HBOT delivers hyperoxia to tissues, and it is angiogenic in normal tissues, but it is not angiogenic in tumor tissues.[9]
Finally, hyperoxia alerts and activates the immune system to target cancer cells, first by MHC-1 antigen-presenting cells as well as specifically cancer-focused cytotoxic T-cells. This effect has been shown to lead to regression of tumors.[10]
The above factors are likely why exercise has been so helpful in successful cancer outcomes. It brings in vastly increased oxygen through the lungs to the tissues.
This is how Nature Works Best Cancer Clinic makes use of vulnerability of cancer cells, and a synergy between IV nutrients and significantly increased amounts of oxygen imparted to cancer cells, which they cannot tolerate, while at the same time nourishing normal cells with oxygen.
Safety
Nature Works Best Medical Clinic decided on soft-sided chambers without additional oxygenation, because of previously reported risks of fires and explositions in high pressure chambers with 100% added oxygen. Although the accidents with those high-pressure chambers are thankfully very rare, the FDA warns about precautions with those chambers.[11]
At the clinic, we do not add supplemental oxygen in the soft-sided chambers, thereby avoiding the fire risk of the chambers with added oxygen and high pressure.
Barotrauma is the only recognized risk with soft-sided chambers. This is similar to driving up or down mountain roads, where the ears “pop” from pressure changes with different alltitude. In these cases we advise patients to yawn (most recommended) or swallow or chew gum, in order to adjust to the higher pressure at the beginning of an HBOT session or the decreasing pressure at the end. Our chambers are pressured during a “dive” (like a scuba dive) to 1.3 ATM, which is 30% more air pressure than at sea level. It has been compared to standing at the shore of the Dead Sea in Israel. Just as when ascending or descending a mountain or on a flight, yawning or swallowing helps to equalize pressure on both sides of the eardrum.
Precautions
Let your healthcare provider know if you have had a recent ear infection, other ear injury, claustrophobia or an implanted device such as a pacemaker. You may need to check with the device manufacturer for information about safe use with a HBOT chamber.
History of hyperbaric therapies
In 1662, Nathaniel Henshaw built the first widely known hyperbaric chamber, which he named The Domicilium, and used it to treat a number of health conditions.
The French physician Fontaine built a pressurized mobile operating room in 1878.
Dr. Orville Cunningham, a professor of anesthesia, built the “Steel Ball Hospital” in Cleveland, Ohio, on the shore of Lake Erie in 1928. It was six stories high and 64 feet in diameter, and treated patients with a variety of conditions, beginning with the successful reversal of influenza. The Great Depression hit the economy so hard that the hospital was later torn down for scrap metal.
As of the present, over 10,000 clinical studies and trials have been conducted for HBOT treatment of a great variety of conditions, with an overwhelming majority reporting consistent success.
– Nature Works Best Medical Clinic
-
X Li, S Carlin, et al. Visualization of hypoxia in microscopic tumors by immunofluoescent microscopy. Aug 2007. Cancer Res. 67 (16). 7646-7653. https://aacrjournals.org/cancerres/article/67/16/7646/533311/Visualization-of-Hypoxia-in-Microscopic-Tumors-by
C Roma-Rodrigues, R Mendes, et al. Targeting tumor microenvironment for cancer therapy. Feb 2019. Int J Mol Sci. 20 (4). 840. https://pmc.ncbi.nlm.nih.gov/articles/PMC6413095/
H Li, M Rokavec, et al Antagonistic effects of p53 and HIF1A on microRNA-34alpha regulation of PPP1R11 and STAT3 and hypoxia-induced epithelial to mesenchymal transition in colorectal cancer cells. Aug 2017. Gastroenterology. 153 (2). 505-520. https://www.gastrojournal.org/article/S0016-5085(17)35510-5/fulltext
P Birner, M Schindl, et al. Overexpression of hypoxia-inducible factor 1 alpha is a marker of unfavorable prognosis in early stage invasive cervical cancer. Sep 2000. Cancer Res. 60 (17). 4693-4696. https://pubmed.ncbi.nlm.nih.gov/10987269/
Y Huang, D Lin, et al. Hypoxia-inducible factor (HIF) in the tumor microenvironment. Oct 2017. Sci China Life Sci. 60 (10). https://pmc.ncbi.nlm.nih.gov/articles/PMC6131113/
X Jing, F Yang, et al. Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Nvo 2019. Mol Cancer. 18 (157). https://pmc.ncbi.nlm.nih.gov/articles/PMC6844052/#CR16
Ibid. Jing, et al.
Q Chen, M Espey, et al. Pharmacologi ascorbic acid concentrations selectively kill cancer cells: Action as a pro-drug to deliver hydrogen peroxide to tissues. Sep 2005. Proc Natl acad Sci. 102 (38). 13604-13609. https://pmc.ncbi.nlm.nih.gov/articles/PMC1224653/
J Folkman. Clinical applications of research on angiogenesis. Dec 1995. NEJM [seminar] 333. 1757-1763. https://www.nejm.org/doi/10.1056/NEJM199512283332608
S Hatfield, J Kjaergaard, et al. Systemic oxygenation weakens the hypoxia and Hypoxia Inducible Factor 1-alpha-dependent and extracellular adenoxide-mediated tumor protection. Aug 2014. J Mol Med (Berl). 92 (12). 1283-1292. https://pmc.ncbi.nlm.nih.gov/articles/PMC4247798/
https://www.fda.gov/medical-devices/letters-health-care-providers/follow-instructions-safe-use-hyperbaric-oxygen-therapy-devices-letter-health-care-providers